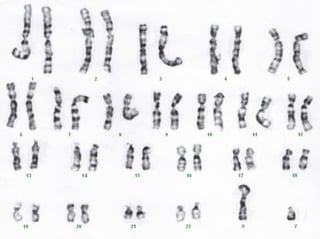
Fetal aneuploidy affects about 9 in 1,000 live births. The definition of aneuploidy is an abnormal number of chromosomes ; with 23 pairs of chromosomes in humans, 46 is the normal number, while aneuploidy individuals will have 45 or 47. In trisomy, there is one additional chromosome, typically chr21, 18 or 13 (it is not a coincidence that these are the smallest chromosomes in humans). Historically, the invasive methods amniocentesis and chorionic villus sampling (CVS) were used with risk to the pregnancy, with about a 1% chance of miscarriage due to the procedure. Non-invasive methods based upon ultrasound and serum biomarkers are useful screening tests, but were of limited reliability as they were indirect measures of chromosomal abnormalities1.
 Photograph courtesy of Flickr user Can H.
Photograph courtesy of Flickr user Can H.
Background
In 1979 it was first reported that maternal plasma contained fetal cells, and in 1990 fetal DNA was detected2. In the ensuing years several technical challenges starting with the relatively high amount of maternal circulating DNA prevented a direct aneuploidy measurement. Fetal circulating RNA and the methylation status of the fetal DNA were both investigated as potential avenues of determining aneuploidy status of the fetus, as well as digital PCR approaches but all suffered from a variety of technical limitations.
The introduction of massively parallel sequencing, the 454 GS20 instrument (acquired by Roche) in 2005, while a huge leap forward in sequencing throughput in bases per day (on the order of 100-fold, compared to the most advanced Sanger sequencing instruments available) did not immediately change the landscape of human genetics research. It took time for new enrichment technology and platforms to develop, to take advantage of this increase in throughput. A few years later in 2007 the Solexa technology (acquired later by Illumina) presented another 10-fold improvement in overall sequencing throughput, but with an important change in sequencing dimensions over the 454 instrument: instead of 1 million 200 base-pair reads, the first Genome Analyzer produced 30 million 36 base-pair reads. This increase in read number (rather than overall sequencing throughput) enabled a new approach to prenatal testing.
The first two papers
In 2008 two important papers were published in the Proceedings of the National Academy of Sciences; the first by Stephen Quake’s group at Stanford University (“Noninvasive diagnosis of fetal aneuploidy by shotgun sequencing DNA from maternal blood”4) and the second by Dennis Lo’s group at The Chinese University of Hong Kong (“Noninvasive prenatal diagnosis of fetal chromosomal aneuploidy by massively parallel genomic sequencing of DNA in maternal plasma”5). Independent of the fetal genotype, a ‘shotgun sequencing’ approach randomly samples millions of sequence tags across the genome, and any over-representation or under-representation of any chromosome representing the fetal portion can be detected above the background maternal signal.
These first two papers conclusively showed the ability to detect fetal trisomy 21 (Down Syndrome), trisomy 18 (Edwards Syndrome) and trisomy 13 (Patau Syndrome) from maternal plasma as a non-invasive test, with sensitivity and specificity at 100% in the limited number of samples they tested. Subsequent larger studies have since been conducted, and a meta-analysis showed5 that these NGS-based non-invasive pre-natal tests (NIPT) were 99% sensitive for Trisomy 21 at 99.9% specificity, 97% sensitive for Trisomy 18 at 99.9% sensitivity, and 92.1% sensitive for Trisomy 13 at 99.8% specificity.
Rapid adoption
Many companies have been founded worldwide to commercialize this technology, and offer this test on an experimental basis. There were three predominant approaches: a massively-parallel shotgun sequencing approach (as described in the first two PNAS publications), chromosome-targeted sequencing, and single nucleotide polymorphism (SNP) genotyping-based analysis. Steven Quake speaking at a conference estimated that in 2014 over 1 million of these tests have been performed worldwide, and said NIPT was the most quickly-adopted molecular test in history.
Since the first publication in 2008 by Steven Quake's group4, the field moved forward rapidly out of the research labs into more routine clinical practice. in 2019, the number of NIPT tests worldwide was approximately 8 million. Changes in professional guidelines8, initiated expansion of the use of NIPT testing beyond the women of advanced maternal age to a low risk population. This increase of test numbers is accompanied by further evolution of testing methods, which are continuously being improved.
SeraCare is committed to insuring the reliability of cell-free fetal DNA detection by offering Seraseq NIPT Reference Materials, including a recently updated portfolio of matched maternal-fetal products. More information about our products can be obtained by contacting us here.
References:
1. Wapner R, Thom E, Simpson JL, Pergament E, Silver R, Filkins K, Platt L, Mahoney M, Johnson A, Hogge WA, Wilson RD, Mohide P, Hershey D, Krantz D, Zachary J, Snijders R, Greene N, Sabbagha R, MacGregor S, Hill L, Gagnon A, Hallahan T, Jackson L; First Trimester Maternal Serum Biochemistry and Fetal Nuchal Translucency Screening (BUN) Study Group. First-trimester screening for trisomies 21 and 18. N Engl J Med. (2003) 349(15):1405-13. PubMed PMID: 14534333.
2. Bianchi DW, Flint AF, Pizzimenti MF, Knoll JH, Latt SA. Isolation of fetal DNA from nucleated erythrocytes in maternal blood. Proc Natl Acad Sci USA (1990) 87(9):3279-83. PubMed PMID: 2333281.
3. Tabor A, Philip J, Madsen M, Bang J, Obel EB, Nørgaard-Pedersen B. Randomised controlled trial of genetic amniocentesis in 4606 low-risk women. Lancet. (1986) 1(8493):1287-93. PubMed PMID: 2423826.
4. Fan HC, Blumenfeld YJ, Chitkara U, Hudgins L, Quake SR. Noninvasive diagnosis of fetal aneuploidy by shotgun sequencing DNA from maternal blood. Proc Natl Acad Sci USA. 2008 105(42):16266-71. doi: 10.1073/pnas.0808319105 PubMed PMID: 18838674.
5. Chiu RW, Chan KC, Gao Y, Lau VY, Zheng W, Leung TY, Foo CH, Xie B, Tsui NB, Lun FM, Zee BC, Lau TK, Cantor CR, Lo YM. Noninvasive prenatal diagnosis of fetal chromosomal aneuploidy by massively parallel genomic sequencing of DNA in maternal plasma. Proc Natl Acad Sci USA. (2008) 105(51):20458-63. doi:10.1073/pnas.0810641105. PubMed PMID: 19073917.
6. Gil MM, Akolekar R, Quezada MS, Bregant B, Nicolaides KH. Analysis of cell-free DNA in maternal blood in screening for aneuploidies: meta-analysis. Fetal Diagn Ther. (2014) 35(3):156-73. PubMed PMID: 24513694.
7. Dondorp W, de Wert G, Bombard Y, Bianchi DW, Bergmann C, Borry P, Chitty LS, Fellmann F, Forzano F, Hall A, Henneman L, Howard HC, Lucassen A, Ormond K, Peterlin B, Radojkovic D, Rogowski W, Soller M, Tibben A, Tranebjærg L, van El CG, Cornel MC. Non-invasive prenatal testing for aneuploidy and beyond: challenges of responsible innovation in prenatal screening. Eur J Hum Genet. 2015 (11):1438-50. doi: 10.1038/ejhg.2015.57. PubMed PMID: 25782669.
8. Gregg AR, Skotko BG, Benkendorf JL, Monaghan KG, Bajaj K, Best RG, Klugman S, Watson MS. Noninvasive prenatal screening for fetal aneuploidy, 2016 update: a position statement of the American College of Medical Genetics and Genomics. Genetics in Medicine 2016 (18):1056-1065. PubMed PMID: 27467454




